Pharmaceutical Packaging
Custom medicine boxes
Pharmaceutical Packaging
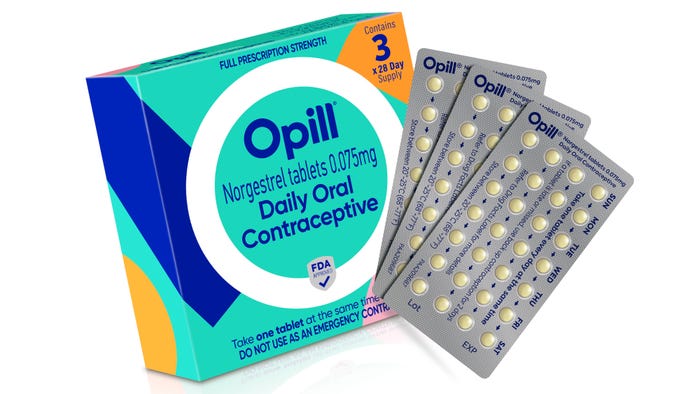
Custom Boxes Elevate Medicine PackagingCustom Boxes Elevate Medicine Packaging
Pharma brand owners can differentiate their offerings using custom packaging that sets them apart from competitors at retail.
Sign up for the Packaging Digest News & Insights newsletter.